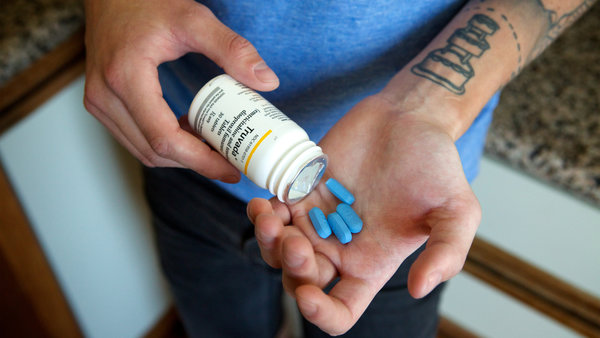
An analysis of blood levels of Truvada (tenofovir disproil fumarate+emtricitabine) among participants in the global iPrEx (Iniciativa Profilaxis Pre-Exposición) study found that U.S. study participants were significantly more likely to take pre-exposure prophylaxis (PrEP) regularly than were their global counterparts. Other indicators of higher adherence to the regimen were older age and riskier sexual practices.
iPrEx was a placebo-controlled phase III trial to determine whether Truvada was effective in preventing HIV infection in men who have sex with men (MSM) and in transgender women. It enrolled 2499 participants in Latin America, Thailand, South Africa and the U.S. between 2007 and 2011. The trial started in the Andes (Peru and Ecuador), so a larger proportion of the overall study population came from that region. Participants in the U.S. comprised 9% of the 1251 people in the iPrEx active arm (i.e., those receiving Truvada rather than the placebo), while Andean MSM and transgender women accounted for 68% of iPrEx active arm participants.
As described in “Patterns and Correlates of PrEP Drug Detection,” by Albert Liu, M.D., M.P.H., et al., researchers conducted a cross-sectional analysis that measured Truvada levels at week 8 in the blood of 470 participants randomly selected from the active arm of the iPrEx trial. It also evaluated a cohort of 303 iPrEx participants over 72 weeks to determine consistency of medication adherence. Seventeen percent of people in the week 8 cohort were at U.S. sites, while 44% were in the Andes. The respective numbers for the 72-week longitudinal cohort were 11% from the U.S. and 56% from the Andes.
Keep reading…






 TrafficHolder.com - Buy & Sell Adult Traffic
TrafficHolder.com - Buy & Sell Adult Traffic
[…] As described in “Patterns and Correlates of PrEP Drug Detection,” by Albert Liu, M.D., M.P.H., et al., researchers conducted a cross-sectional analysis that measured Truvada levels at week 8 in the blood of 470 …read more […]
[…] Analysis of PrEP Data Shows High Adherence in U.S., and Among Most at Risk of HIV […]